educational resources
CDMRP Hearing Restoration Resources for Investigators
Hearing Restoration
Vision – Reduce the burden of hearing loss on Service Members, Veterans, and the American public
The HRRP was initiated in 2017 to pursue promising, necessary research for treatment of burdensome and very prevalent auditory system injury. It is estimated that more than 30 million Americans over the age of 12 years have hearing loss in both ears and an estimated 48 million have hearing loss in at least one ear. In the military the two most prevalent service-connected disabilities are related to hearing disorders. The most recent data from the Veterans Benefits Administration, Department of Veterans Affairs indicates that there are 1.1 million Veterans with service-connected disability due to hearing loss. The HRRP will fund innovative research that has the potential to maximize operational effectiveness, medical readiness and quality of life for Service members, Veterans and others living with significant auditory system injuries.
The Latin American Neurotology Journal Club

The Latin American Neurotology Journal Club, was created in 2020 as an initiative by two otolaryngologists to keep updated on field related news and have virtual academic meetings with colleagues. Known neurotologists and other professionals from Latin America are members of this group. Currently, this group is the first academic space for articles discussion and academic exchange between professionals from different areas related to Neurology and Vestibular disorders in Spanish.
Every meeting, one article of a different topic is discussed. Topics are elected by poll and all members read the article for general discussion. Additionally, other specialists, such as Endocrinologists, Gynecologists and Neurologists are invited for some meetings in order to solve questions about related topics and complimentary management of some diseases (i.e D-vitamin use and treatment in benign paroxysmal positional vertigo). From the discussions, ideas related to collaborations and research have been emerged. Some members have also shown their current on-course research works to the rest of the group to have feedback from other colleagues around the region.
The Latin American Neurotology Journal Club meets every third Wednesday at 8:30 PM (EST) via Zoom.
For questions and to get involved, please contact ARO/spARO Member, Melissa Castillo-Bustamante at Melissa_castillobustamante@meei.harvard.edu
Vestibular Cafe Podcast
The Vestibular Cafe is one podcast led by Latin American healthcare professionals and researchers focused on highlighting the journey and challenges of healthcare providers dedicated to balance and vestibular disorders
The podcast talks about several topics on leadership, mentoring, challenges on healthcare attention and research but on an easygoing way. This podcast is led by Jorge Madrigal, MD Otoneurologist (currently applying to ARO Membership) and Researcher in Mexico City, Fabricio Martínez MD, Otoneurologist in Buenos Aires and Melissa Castillo-Bustamante MD, Otolaryngologist, Researcher and Member of the Membership committee at ARO
Our aim is to connect experiences in leadership and research of Latin American healthcare professionals and those who are currently practitioners and future colleagues
ARO 2020 Photo Contest
Below you will find stunning images from the research labs of our members, submitted in response to an ARO Art Contest held in January 2020. These images can be seen scattered throughout our web pages. Please make sure that you do not use these images without crediting the source. Thank you & enjoy!
First Place
Credit: Patrick Lam, Ella Trang, Niki Gunewardene, and Andrew Wise of Bionics Institute
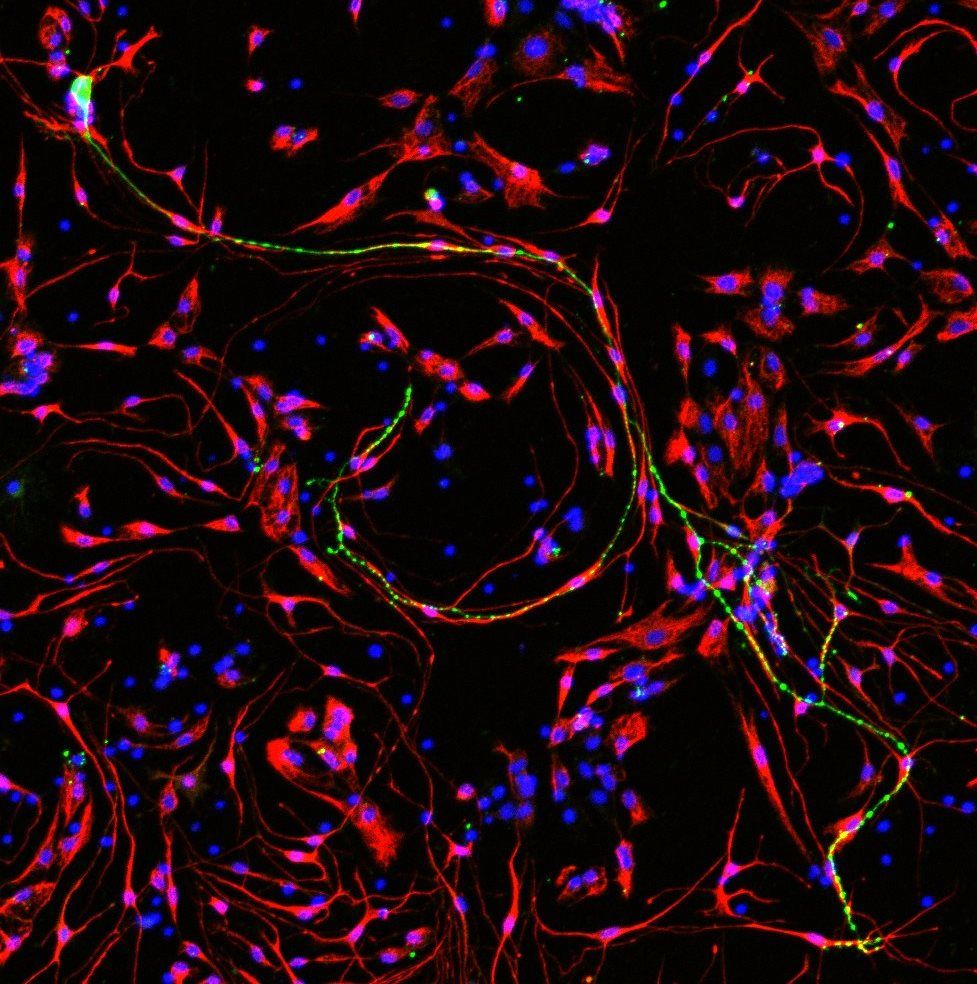
Description: 3-day culture of primary spiral ganglion cells isolated from 5D neonatal Sprague Dawley rats.This image shows a common phenomenon of spiral ganglion neuron processes being surrounded by glial cells that could play an important role in the nerve survival and signalling.Cells were visualised via immunocytochemistry. Labelled in green are two spiral ganglion neurons using Tuj1 as the primary antibody binding marker; in red are the glial cells using S100 beta as the primary antibody binding marker; all cell nuclei were stained with DAPI in blue.
Second Place
Credit: David Bächinger, Andreas H. Eckhard
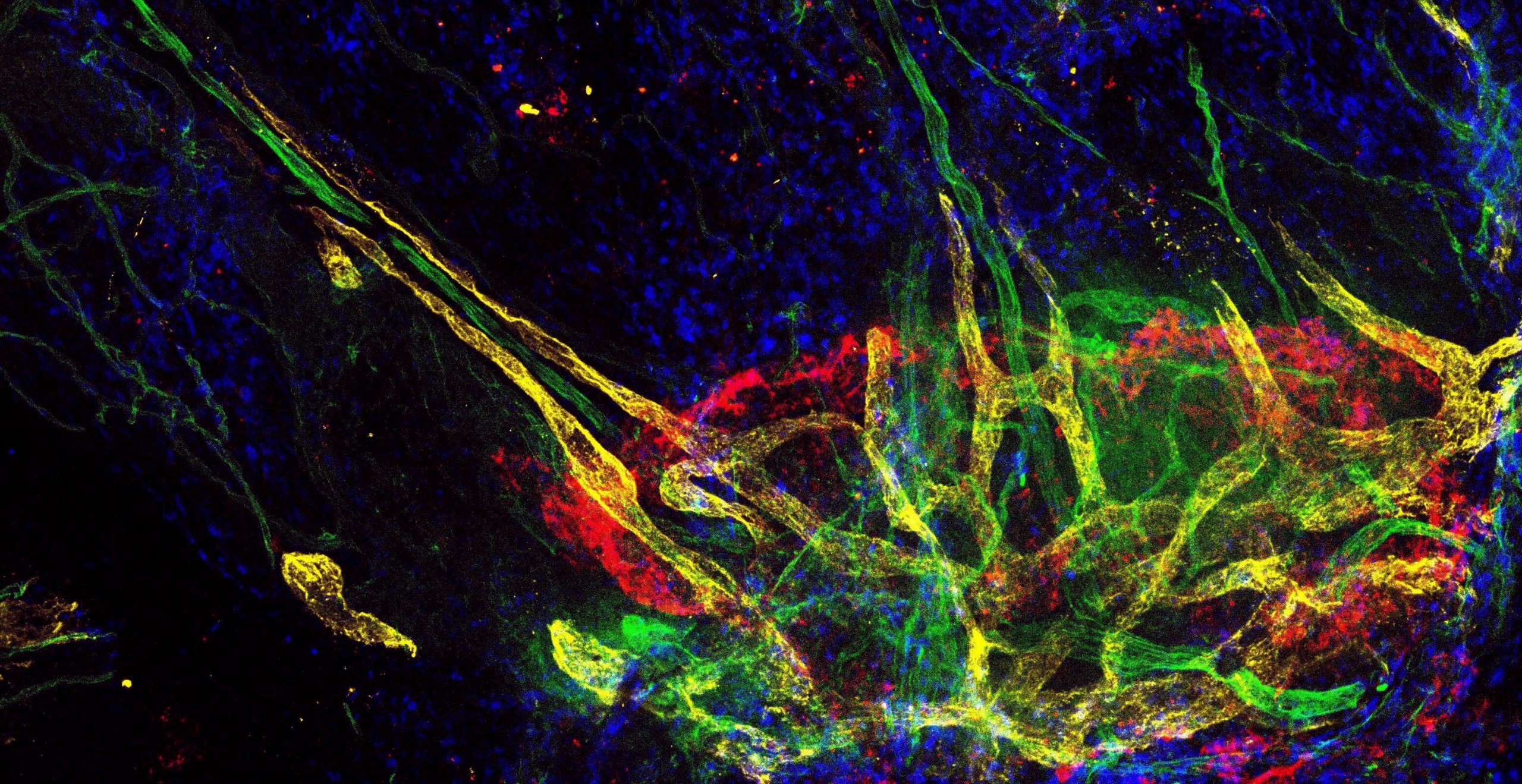
Description: This image shows a quadruple immunostaining a of a whole-mount specimen of the adult murine endolymphatic sac (6 weeks old female C57BL/6J). Lymphatic vessels were stained with an anti-LYVE1 antibody (yellow). Concanavalin A (green) was used to mark blood vessels. The endolymphatic sac is outlined by epithelial staining using a pan-cytokeratin antibody (red). Cell nuclei are marked with DAPI (blue). Note that lymphatic (yellow) and blood vessels (red) form a “star-like” hub in the endolymphatic sac region
Third Place
Credit: Leonardo R. Andrade of Salk Institute for Biological Studies and NIDCD/NIH
Description: SEM image of bullfrog otoconia.